| Humulin Regular | |
|---|---|
| R, Neutral by Eli Lilly | |
| short-acting | |
| r-DNA/GE/GM | |
| U40, U100 | none |
Action in dogs:
| |
| Line: Humulin | |
| Also known as: Huminsulin Normal Humulin C, Humulin R, Humuline R Humulina Regular, Humuline Regular Humulin S, Humulin ReliOn R, Umuline Rapide Umuline Regular | |
| Similar to: Insuman Rapid, Winthrop Rapid Actrapid, Novolin R, ReliOn/Novolin R Names of Lilly r-DNA/GE/GM insulins worldwide | |
| Use and Handling: | |
| Shelf Life: 24 months | Type: soluble, clear |
| When Opened: 28 days room temp. | |
| In Pen: 28 days room temp. | |
| Notes: Protect from light and heat Do Not Freeze, May be used intravenously | |
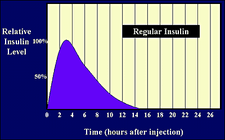
Humulin Regular time activity profile.
|
Note that some countries have the brand name in U40 strength. | ||
Humulin Regular is one of several brands of "Regular" (short-acting) insulin, used for a quick consumption of blood sugar with little delay.
It consists of zinc insulin crystals dissolved in clear fluid with no additives to alter its action or speed. [2]
Other brands with the same action profile include:
- Novolin R [3]
- Humulin S [4][5][6]
- Huminsulin Normal
- Actrapid [7][8][9]
- Insuman Rapid [10][11].
- Winthrop Rapid
A short-acting, soluble, non-suspended insulin produced from r-DNA/GE/GM, bovine, or porcine sources. From 1921 to the 1930's when PZI was developed, R was the only kind of insulin available.
While both R/neutral and semilente insulins are considered fast-acting, they differ in a few ways. As above, there's no suspension or buffer [12] in R/neutral; it's totally dissolved and soluble, meaning it can safely be used intravenously. [13]
The very small insulin particles in semilente insulin are suspended by zinc. [12] Semilente is cloudy, has an acetate buffer and cannot be used intravenously.
R/Neutral insulin cannot be reliably mixed with Lente or Ultralente insulins. [14][15] Semilente can; the ratio of 3 parts semilente insulin to 7 parts ultralente insulin produces Lente insulin. [12]
R/Neutral Insulins[]
|
R/Neutral Insulins | |
|---|---|

|
All Short acting All soluble |
| Hypurin Bovine Neutral Betasint Bovine Corriente | |
| Iletin R (No longer produced.) Hypurin Vet Neutral (No longer produced.) Insuvet Neutral (No longer produced.) | |
| Iletin I R (No longer produced.) | |
| Betasint Porcine Corriente Hypurin Porcine Neutral Hypurin Pork Regular Maxirapid CHO | |
| Pork Actrapid (No longer produced.) Iletin II R (No longer produced.) Leo Neutral (No longer produced.) | |
| Actrapid, Huminsulin Normal Humulin C, Humulin R Humulin Regular, Humulina Regular Humuline R, Humuline Regular Humulin S, Insuman Rapid Novolin R, Umuline Rapide Umuline Regular, Winthrop Rapid Humulin ReliOn R | |
| R-DNA | ReliOn/Novolin R (No longer produced.) |
References[]
- ↑ Insulins. North American Veterinary Conference Clinicians Brief (2006).
- ↑ Humulin R patent leaflet. Eli Lilly.
- ↑ Novolin R Regular Human Insulin. Revolution Health.
- ↑ Patient Information Leaflet-Humulin S. Eli Lilly.
- ↑ Humulin S Prescribing Information
- ↑ Lilly EU/UK Product Sheet & Time Activity Profiles. Eli Lilly.
- ↑ Patient Information Leaflet-Actrapid. Novo Nordisk.
- ↑ Scientific Discussion-Actrapid. EMEA.
- ↑ Prescribing Novo Insulins.
- ↑ Patient Information Leaflet-Insuman Rapid. Sanofi-Aventis.
- ↑ Prescribing Insuman Insulins.
- ↑ 12.0 12.1 12.2 DeRuiter, Jack. Insulin Preparations-Regular Insulin. Auburn University. Cite error: Invalid
<ref>tag; name "Auburn" defined multiple times with different content Cite error: Invalid<ref>tag; name "Auburn" defined multiple times with different content - ↑ Maddison, Jill E.,Page, Stephen W.,Church, David B. (2008). Small Animal Clinical Pharmacology. Saunders Ltd..
- ↑ Deckert, T. (1980). Intermediate-Acting Insulin Preparations: NPH (Isophane) & Lente. Diabetes Care.
Note--in 1980, there were no r-DNA/GE/GM insulins - ↑ Resource Guide. American Diabetes Association (2005).
More Information[]
- NACDS-Insulin Chart-Page 2
- Leeds NHS UK Insulin Guide
- Insulin therapy for dogs and cats Dowling, Patricia, September 1995, Canadian Veterinary Journal
A discussion of regular/neutral/normal insulins.
| Related Pages | ||||
|---|---|---|---|---|
|

|

|

|

|